Key Message:
- There is a scarcity of N95 masks and other PPE for doctors
- They need a method to decontaminate the face masks to reuse
- The best method to make the N95 sterile is: Place the mask or even PPE in a laminar flow hood and turn on UV light at maximum (220-254nm). To be on the safe side, irradiate for 2h
- Other reported methods on the web, google don’t work because they compromise the HEPA filter and the masks
Right now our doctors are struggling to have N95 masks and other PPE (personal protective equipment). These masks are made to be used once and thrown. However, if the seriousness of circumstances dictates reuse of the masks, we need a method to decontaminate them without compromising the durability of the masks.
Several methods are coming out ranging from washing with bleach to hydrogen peroxide, autoclaving, boiling, etc. These methods cannot work even if correct temperatures to deactivate the virus are used because these methods break the masks, compromise the HEPA filters.
As a Geneticist who has worked with viruses for years, the best method to decontaminate the N95 masks is as follows:
In the laminar flow hood or a sterile hood, place the mask or even PPE and turn on UV light at maximum (220-254nm). To be on the safe side, let the paraphernalia stay under UV for 2h. This is the method we use in the research labs to decontaminate areas from viruses. Ultraviolet light has been used in the research laboratory as an effective germicide and virucide.
Germicidal lamps emit radiation almost exclusively in the far-UV range of 254 nm and are commonly used in Laminar Air Flow hoods or biological safety cabinets. The inverse square law applies to the UV light as it does to light: the killing power of UV decreases as the distance from the lamps increases. The average bacterium/virus will be killed in ten seconds at a distance of six inches from the lamp in an American Ultraviolet Germicidal Fixture.
Ultraviolet light is, by definition, light with shorter wavelengths and is generally broken down into three distinct subgroups:
- UVA, consisting of light with a wavelength between 400 and 320 nm
- UVB, consisting of wavelengths between 320 and 290 nm; and
- UVC, with wavelengths between 290 and 200 nm.
For disinfection purposes, the optimal wavelengths reside within the UVC range and low-pressure mercury lamps provide a nearly monochromatic 254.6 nm output. The UV-C is the active part of the light. UV inactivation doses have been determined for a variety of organisms. UV light has been found to be a fairly efficient disinfectant for most vegetative organisms and viruses.
UV light has even been shown to kill 99.9 percent of the molds over a one to two hours period. Even at the minimum acceptable irradiance in a biosafety cabinet of 40 W/cm2 (U.S. Department of Health and Human Services et al., 2000), it takes 12.5 minutes to reach 30,000 J/cm2 (1 W = 1 J/sec), which has been listed as germicidal for spore-forming organisms.
D37 values (fluence producing on average one lethal hit per virion and reducing viable virus to 37%) have been calculated for several viruses. “Size-normalized sensitivity” (SnS) is defined by multiplying UV254 sensitivities (D37 values) by the genome size. SnS values have been found to be relatively constant for viruses with similar genetic composition.
Besides, SnS values are similar for complete virions and their defective particles, even when the corresponding D37 values might be significantly different. SnS values have been used to estimate the UV254 sensitivities of viruses for which the genome composition and size are known but no UV inactivation data are available, e.g. smallpox virus, Ebola, Marburg, Crimean-Congo, Junin, and other hemorrhagic viruses, and Venezuelan equine encephalitis and other encephalitis viruses.
How does UV light kill the viruses and bacteria
DNA (Deoxyribonucleic acid) is the critical target for killing by UV light. The nucleic acid within the virus particle plays a crucial role in the absorption of UV radiation thus inactivates viruses. Nucleic acids are also damaged by UVB and UVA but with lower efficiency than by UVC radiation (64). Two major adducts are formed by UV light, both of which are bimolecular adducts of pyrimidine bases.
UV targets the thymines bases in the viruses. Since DNA contains thymine, but not RNA, DNA-containing viruses are more sensitive to damage by UV than RNA-containing viruses. Also, the viruses that possess double-stranded nucleic acids can repair their nucleic acids, thus reduce the lethal effect of UV.
The number of bases in the DNA or RNA is important for determining sensitivity to UV inactivation, because the more target molecules, the more likely the genome will be damaged at a given level of UV exposure.
In most viruses, the other major constituents of the virus particles play relatively minor roles in inactivation by UV (55). For example, whole MS2, f2, Q, encephalomyocarditis virus (EMCV), and murine polyomaviruses and their respective free nucleic acids have essentially the same UV254 sensitivities. The loss of viral infectivity of MS2 in the presence of UV light was found to be due to RNA damage.
Can Coronavirus-19 be deactivated by UV?
COVID-19 is a single-stranded RNA virus. The only difference between DNA and RNA is that DNA has thymine (UV targets that), and RNA has Uracil instead of thymine. However, UV light also makes uracil dimers and other RNA photoproducts from adjacent pyrimidine nucleotides. It also makes RNA-protein cross-links.
These lesions
- inhibit RNA synthesis primarily through the formation of 6-4 photoproducts
- block the reverse transcriptase enzyme from transcribing strands of cDNA
- thus make the reverse transcription-quantitative PCR (RT-qPCR) assay suitable for detecting damage to the viral RNA.
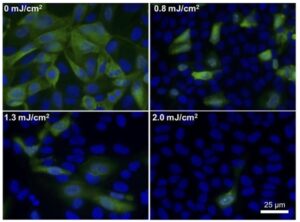
Recently, it has been shown for the first time that far-UVC efficiently inactivates airborne aerosolized viruses, with a very low dose of 2 mJ/cm2 of 222-nm light. This inactivates >95% of aerosolized, H1N1 influenza virus.
Continuous very low dose-rate far-UVC light in indoor public locations could be a promising, safe and inexpensive tool to reduce the spread of airborne- mediated microbial diseases (I will discuss this in another post after I have more data).
Viral Proteins Play an Important Role in Mechanism of Inactivation
Adenovirus is a UV-resistant pathogen while MS2 (Male Specific Coliphage) can be easily inactivated with UV. The difference lies in the differences in their viral proteins. In the case of MS2, maturation protein exists as a single copy. It infects by attaching its maturation protein to the viral receptor on the F pilus of E. coli.
However, Adenovirus has a more complicated protein structure for infecting human cells. It consists of 13 structural proteins, which play an integral role in the attachment, entry, and release of DNA into the host cell’s nucleus.
We got lucky in the sense that Coronavirus-19 doesn’t have a complex protein structure surrounding the RNA. Its genome encodes four major structural proteins:
- spike (S) protein with which it attaches to the host cell receptors thus fusing with host cell membranes
- nucleocapsid (N) protein: binds to CoV RNA and makes the nucleocapsid
- membrane (M) protein: it is a structural protein, is very abundant and defines the shape of the viral envelope
- envelope (E) protein: smallest protein but most enigmatic and very important. It is localized at the site of intracellular trafficking (the ER, Golgi, and ERGIC), where it participates in CoV assembly and budding. It has been shown that if the CoV lack this protein, they have significantly reduced viral titers and crippled viral maturation. This means they yield viruses that cannot propagate further.
All of the above proteins are required to produce a structurally complete viral particle in the case of CoV2.
Summary
Yes, Coronavirus-19 can be deactivated by UV light. This is a protocol only for the decontamination of Face Masks, PPE and objects. No humans should place themselves or any part of their body in this light because it is hazardous to DNA and will give cancer to the exposed organs.